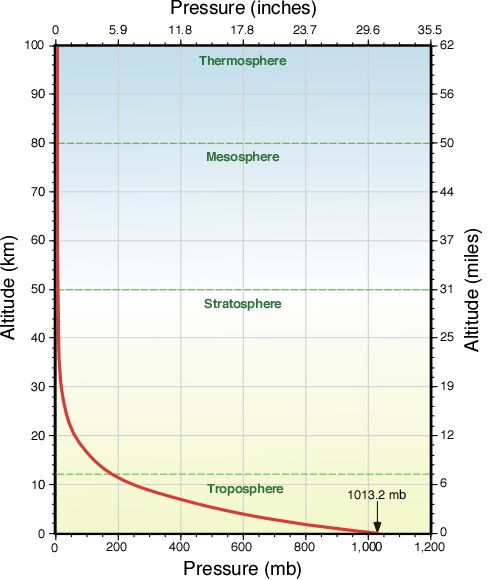
The earth's atmosphere exerts a force on everything within it. This force, divided by the area over which it acts, is the atmospheric pressure. The atmospheric pressure at sea level has an average value of 1,013.25 millibars. Expressed with other units, this pressure is 14.7 lb per square inch, 29.92 inches of mercury, or 1.01 × 105 pascals-this is commonly referred to as 1 atmosphere which is equivalent to placing a 1kg mass(10N weight) on an area of 1 centimeter squared.
Atmospheric pressure decreases with increasing altitude: it is half of the sea level value at an altitude of about 3.1 mi (5 km) and falls to only 20% of the surface pressure at the cruising altitude of a jetliner. Atmospheric pressure also changes slightly from day to day as weather systems move through the atmosphere.
How much pressure are you under?
Earth's atmosphere is pressing against each square inch of you with a force of 1 kilogram per square centimeter (14.7 pounds per square inch). The force on 1,000 square centimeters (a little larger than a square foot) is about a ton!
Why doesn't all that pressure squash me?
Remember that you have air inside your body too, that air balances out the pressure outside so you stay nice and firm and not squishy.
The Natural Pressure within our bodies is also about 1 atmosphere.As such, the internal pressure of our bodies is able to balance out the atmospheric pressure that acts on us.If this natural pressure was not present within our bodies, we would be crushed to death by the atmosphere.
Air pressure can tell us about what kind of weather to expect as well. If a high pressure system is on its way, often you can expect cooler temperatures and clear skies. If a low pressure system is coming, then look for warmer weather, storms and rain.
What Happens if Air Pressure Changes?
Our bodies are unable to adjust quickly to changes in atmospheric pressure, and we may suffer injuries or altitude sickness if the changes are too great.
For Example
We know that air pressure in High altitudes is much lower than 1 atmosphere.Hence, to safeguard the passengers on an aeroplane, the pressure in the aircraft is gradually increased as the plane ascends.This way, the people on board will not experience a great pressure change.
Why do my ears pop?
If you've ever been to the top of a tall mountain, you may have noticed that your ears pop and you need to breathe more often than when you're at sea level. As the number of molecules of air around you decreases, the air pressure decreases. This causes your ears to pop in order to balance the pressure between the outside and inside of your ear. Since you are breathing fewer molecules of oxygen, you need to breathe faster to bring the few molecules there are into your lungs to make up for the deficit.
As you climb higher, air temperature decreases. Typically, air temperatures decrease about 3.6° F per 1,000 feet of elevation.

Simple Pressure Related Applications:
DRINKING STRAW:
A drinking straw is used by creating a suction with your mouth. Actually this causes a decrease in air pressure on the inside of the straw. Since the atmospheric pressure is greater on the outside of the straw, liquid is forced into and up the straw.

SIPHON:
With a siphon, water can be made to flow "uphill". A siphon can be started by filling the tube with water (perhaps by suction). Once started, atmospheric pressure upon the surface of the upper container forces water up the short tube to replace water flowing out of the long tube.
Barometer

Any instrument that measures air pressure is called a barometer. The first measurement of atmospheric pressure began with a simple experiment performed by Evangelista Torricelli in 1643.
In his experiment, Torricelli immersed a tube, sealed at one end, into a container of mercury (see Figure 7d-2 below). Atmospheric pressure then forced the mercury up into the tube to a level that was considerably higher than the mercury in the container.
Torricelli determined from this experiment that the pressure of the atmosphere is approximately 30 inches or 76 centimeters (one centimeter of mercury is equal to 13.3 millibars). He also noticed that height of the mercury varied with changes in outside weather conditions.

Atmospheric pressure is not expressed in terms of pascal(PA) but as the height of the mercury column in the barometer.We can express 1 atmosphere as 760 mm Hg or 76 cm Hg(Hg is the chemical symbol for mercury).To convert mm Hg to Pascals, we simply convert mm Hg to 10(to the power of -3) Hg, and multiply the density of mercury in kg/cubic meter and the gravitational field strength,g in N/Kg.
Manometers
Manometers measure a pressure difference by balancing the weight of a fluid column between the two pressures of interest. Large pressure differences are measured with heavy fluids, such as mercury (e.g. 760 mm Hg = 1 atmosphere). Small pressure differences, such as those experienced in experimental wind tunnels or venturi flowmeters, are measured by lighter fluids such as water (27.7 inch H2O = 1 psi; 1 cm H2O = 98.1 Pa).
To calculate the pressure indicated by the manometer, enter the data below. (The default calculation is for a water manometer with a 10 cm fluid column, with the answer rounded to 3 significant figures.):

Equations used in the Calculation
The pressure difference between the bottom and top of an incompressible fluid column is given by the incompressible fluid statics equation,

where g is the acceleration of gravity (9.81 m/s2).
Asking questions are genuinely good thing if you are not understanding anything completely,
ReplyDeletebut this article presents nice understanding yet.
my homepage; cellulite treatment ventura ca
I pretty much copied this entire page for homework.
ReplyDelete